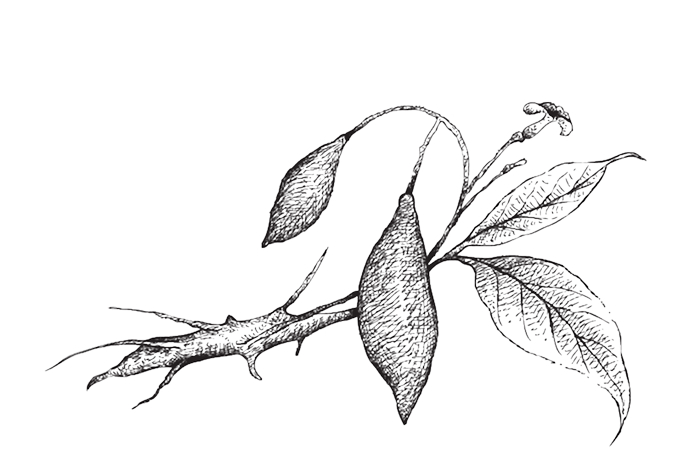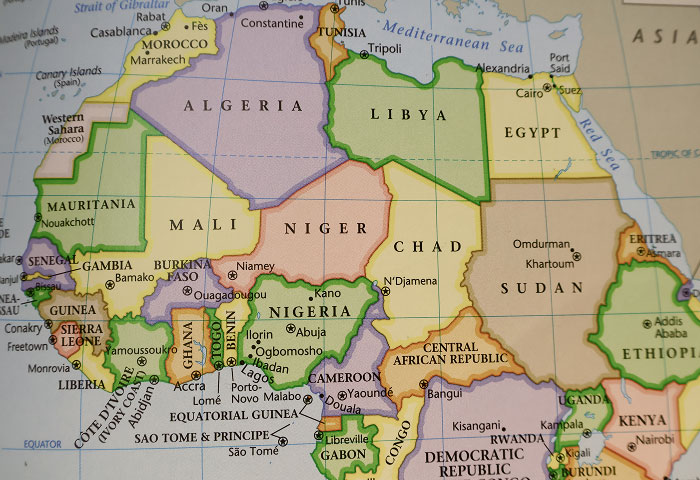
Where Ibogaine Comes From
Ibogaine is a naturally occurring psychoactive compound found in the root bark of Tabernanthe iboga, a shrub native to Central and West Africa. For centuries, the Bwiti tradition of Gabon and Cameroon has used iboga in spiritual and healing ceremonies, a practice that continues today.
Western scientists first isolated ibogaine in 1901, but its potential for treating addiction wasn’t recognized until the 1960s, when anecdotal reports emerged of its ability to interrupt opioid dependence. Since then, a growing body of clinical research has explored ibogaine’s applications for substance use disorders, PTSD, depression, and traumatic brain injury.
How Ibogaine Works
Ibogaine is pharmacologically unique. Unlike conventional addiction treatments that manage symptoms or substitute one dependency for another, ibogaine appears to work on multiple brain systems simultaneously:
Neurological Reset
Research suggests ibogaine interacts with several neurotransmitter systems — including serotonin, dopamine, and glutamate pathways — in ways that may help 'reset' neural circuits disrupted by chronic substance use or trauma.

Withdrawal Interruption
Clinical observations consistently report that ibogaine can significantly reduce or eliminate opioid withdrawal symptoms, often within hours of administration. This effect can create a critical window for patients to engage in therapeutic work without the physical burden of withdrawal.
Neuroplasticity
Emerging evidence indicates ibogaine and its metabolite noribogaine may promote neuroplasticity — the brain's ability to form new neural connections. This is particularly relevant for patients with traumatic brain injury (TBI) and conditions involving cognitive impairment.

Psychological Processing
Many patients report a period of deep introspection during ibogaine treatment that provides insight into the emotional and psychological roots of their addiction or trauma. While subjective, clinicians have noted these experiences often correlate with sustained behavioral change.
Conditions Under Study
Research is actively exploring ibogaine’s potential for:

Ibogaine in the United States
Ibogaine is currently classified as a Schedule I controlled substance in the United States, meaning it is not approved for medical use. Americans for Ibogaine is working to change this through the FDA approval pathway — supporting clinical trials, informing policymakers, and building the evidence base necessary for regulatory action.
Several states have taken legislative action to explore ibogaine research and access. Texas has allocated $50 million for FDA-supervised clinical trials — the largest state investment in ibogaine research in U.S. history.